Availability of Information Given to Advisory Committee Members in Connection with CDRH Open Public Panel Meetings; Draft Guidan
March 28-29, 2023 Joint Meeting of the Drug Safety and Risk Management Advisory Committee and Dermatologic and Ophthalmic Drugs
Amylyx Pharmaceuticals Announces Posting of Briefing Documents for FDA Advisory Committee Meeting on AMX0035
FDA ADVISORY COMMITTEE BRIEFING DOCUMENT MNK-812 JOINT MEETING OF THE ANESTHETIC AND ANALGESIC DRUG PRODUCTS ADVISORY COMMITTE
FDA Briefing Document - Spark Therapeutics, Inc, LUXTURNATM: Cellular, Tissue, and Gene Therapies Advisory Committee Meeting - O
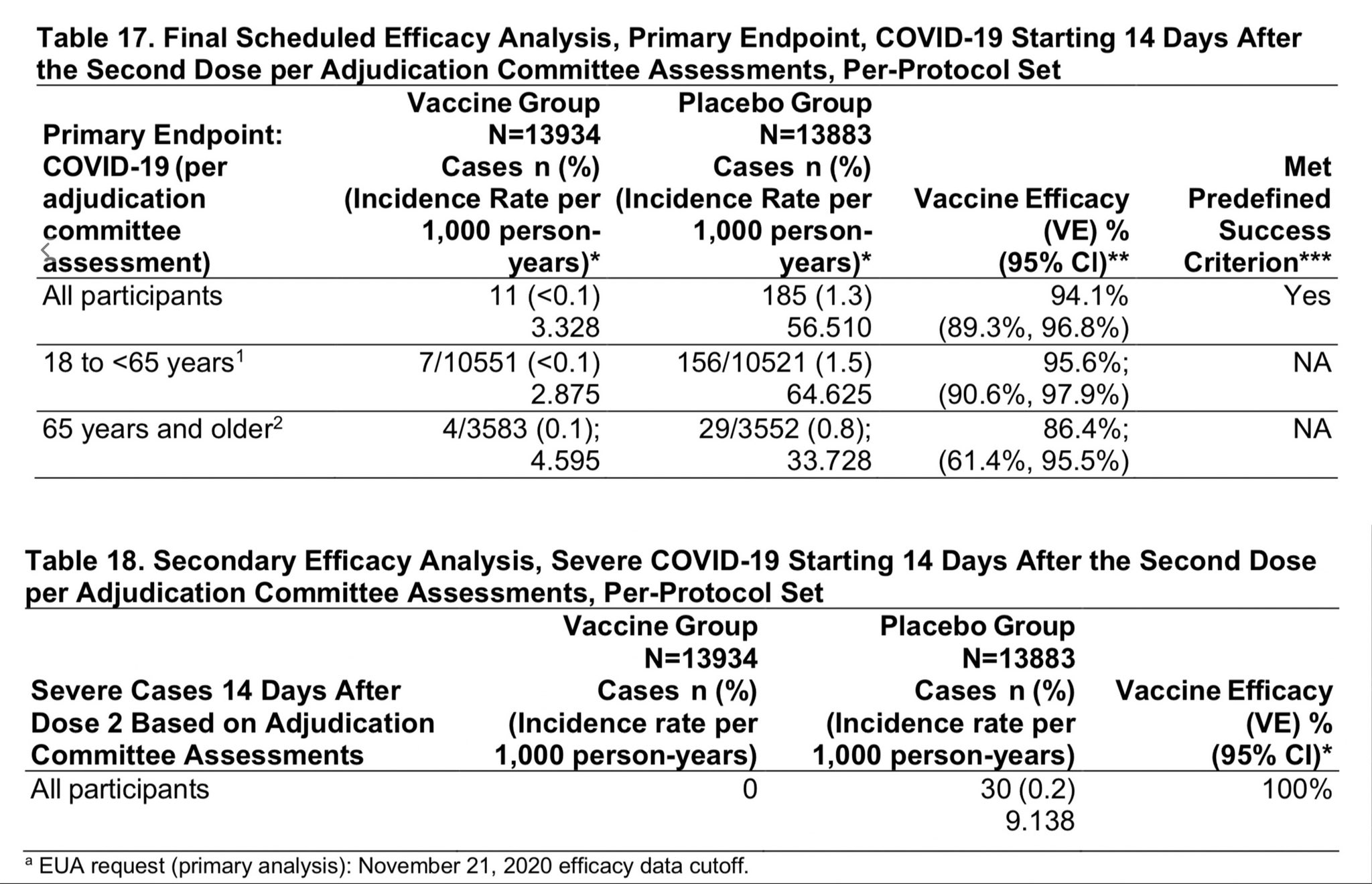
David R. Liu on Twitter: "Want to review #SARSCoV2 #COVID19 vaccine safety and efficacy data yourself? Linked below is the briefing document for the FDA vaccine advisory committee on Moderna's mRNA vaccine,

THE DEBRIEF ON BRIEFING DOCUMENTS: HOW TO MAXIMIZE YOUR BRIEFING BOOK FOR FDA ADVISORY COMMITTEE MEETINGS

SIDP on Twitter: "This just in: FDA advisory committee recommends approval of omadacycline for treatment of ABSSI & CABP. Briefing document here: https://t.co/5y9d8qLUdN https://t.co/aT7Weeu0rI" / Twitter

FDA Lies About PFIZER's mRNA Vaccine Ingredients and Safety in Government Briefing Documents and Website
FDA Briefing Document Joint Meeting of Anesthetic and Analgesic Drug Products Advisory Committee and Drug Safety and Risk Manage
Secukinumab (AIN457) ADVISORY COMMITTEE BRIEFING MATERIAL: AVAILABLE FOR PUBLIC RELEASE Prepared by Novartis Pharmaceuticals Co
1 FDA Briefing Document NDA 216951 Drug name: Daprodustat Applicant: GlaxoSmithKline (GSK) Intellectual Property (No. 2) Limited
March 28-29, 2023 Joint Meeting of the Drug Safety and Risk Management Advisory Committee and Dermatologic and Ophthalmic Drugs

Fillable Online FDA Briefing Document Oncologic Drugs Advisory Committee ... Fax Email Print - pdfFiller
Amylyx Pharmaceuticals Announces Posting of Briefing Documents for Second FDA Advisory Committee Meeting on AMX0035
![PDF] Ziprasidone Mesylate for Intramuscular Injection 1 Advisory Committee Briefing Document Appendix 1, The Behavioural Activity Rating Scale | Semantic Scholar PDF] Ziprasidone Mesylate for Intramuscular Injection 1 Advisory Committee Briefing Document Appendix 1, The Behavioural Activity Rating Scale | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/608dde6ea19d8f7b35c9876408fad41316408421/6-Table2-1.png)




